Technology
CO Utilization Technology
Process and equipment design to solve problem associated with gas emissions
CO utilization for commercial purposes such as NaHCO production would be more beneficial to promote the deployment of CO capture technologies. Our optimized design of the reaction column is able to generate NaHCO from CO without any fouling problem.
Benit M offers the design package for CO utilization
Valuable product of CO utilization
Special tower internal design
No fouling
Carbonation Column
The presence of CO in our daily life cannot be neglected. CO itself has been well-known as the versatile material for generating various products such as dry ice, supercritical fluid solvent, methanol, urea, and etc. However, as the CO level in atmosphere is continuing to rise, more significant efforts must be made to decrease the CO level cost-effectively.
The presence of CO in our daily life cannot be neglected. CO itself has been
well-known as the versatile material for generating various products such as dry ice,
supercritical fluid solvent, methanol, urea, and etc. However, as the CO level in
atmosphere is continuing to rise, more significant efforts must be made to decrease
the CO level cost-effectively.
In particular to remove the CO from industrial flue gas, CO mineralization process
demonstrated greater CO reduction impact compared to other CO utilization technologies.
As one of the CO mineralization process
technology, CO from flue gas can be utilized directly to produce NaHCO.

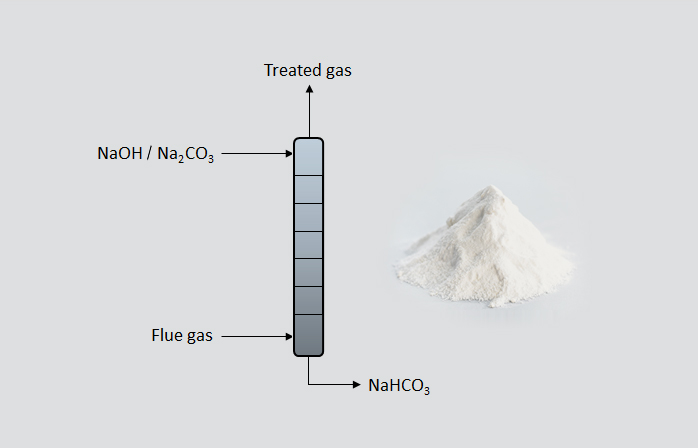
Carbonation Column
NaHCO (sodium bicarbonate), known as baking soda has numerous applications such as food, neutralization of acids, deodorizers, pest control, medical uses, cosmetics, or cleaning agents. NaHCO is produced by reacting CO with NaOH / NaCO in the carbonation column. The carbonation column acts as the CO capture and NaHCO production reactor by following the reactions below.
In particular to remove the CO from industrial flue gas, CO mineralization process
demonstrated greater CO reduction impact compared to other CO utilization technologies.
As one of the CO mineralization process
technology, CO from flue gas can be utilized directly to produce NaHCO.
NaHCO (sodium bicarbonate), known as baking soda has numerous
applications such as food, neutralization of acids, deodorizers, pest control,
medical uses, cosmetics, or cleaning agents. NaHCO is produced by reacting CO
with NaOH in the carbonation column. The carbonation column acts as the CO capture
and NaHCO production reactor by following the reactions below.
NaOH + CO → NaCO + HO
NaCO + CO + HO → NaHCO




















